Superoxide dismutase is most powerful body antioxidant
Superoxide dismutase - an antioxidant enzyme produced by every cell in the body that reduces potentially harmful oxygen free radicals produced during normal metabolic activity of a cell.
The role of superoxide dismutase in the body
Superoxide dismutase (SOD) converts O₂- superoxide anion into H₂O₂ hydrogen peroxide.

1 superoxide dismutase molecule can neutralize up to 1 million free radicals, while vitamin C neutralizes 1 free radical.
Superoxide dismutase (SOD) is a very powerful antioxidant due to its enzymatic ability to neutralize free radicals. This helps to fight oxidative stress and reduces inflammation, slows down the aging process.
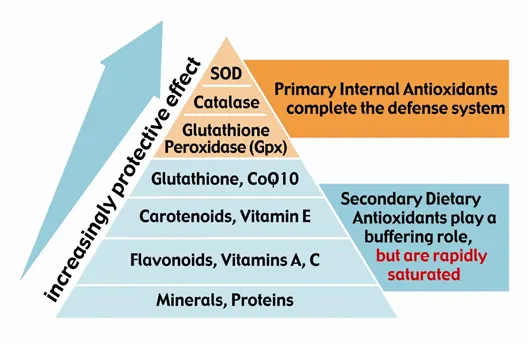
Superoxide dismutase helps regulate the production of antioxidant enzymes, including catalase (CAT) and glutathione peroxidase (GPx), which are required to neutralize reactive oxygen species (ROS).
Application of superoxide dismutase in medicine
Dermatology and Aesthetic Medicine
Superoxide dismutase can reduce free radical damage to skin cells - for example, reduce post-radiation fibrosis in breast cancer. However, studies of this type should be considered preliminary because the study lacked adequate control, including no randomization or placebo. Superoxide dismutase is known to reverse fibrosis, possibly by de-differentiating myofibroblasts back into fibroblasts.
Superoxide dismutase is also capable of protecting skin cells from damage by ultraviolet rays in photodermatosis and is successfully used in the treatment of skin hyperpigmentation. As an adjuvant therapy, it increases the effectiveness of topical treatments and laser treatments in the treatment of hyperpigmentation and melasma.
Positive results were also obtained in the treatment of vitiligo with GliSODin superoxide dismutase drugs.
Superoxide dismutase and aging
Superoxide dismutase is considered an anti-aging enzyme. The free radical theory of aging was proposed by Derham Harman. He suggested that free oxygen radicals generated in metabolic pathways lead to age-related deterioration due to oxidative damage to biomolecules, with mitochondria being the main target of attack. The accumulation of oxidative damage is considered one of the key mechanisms of aging. Drosophila flies, which show a 75% decrease in superoxide dismutase activity, showed an accelerated loss of olfactory behavior with aging. It has been suggested that new superoxide dismutase mimetics may be useful in alleviating cognitive impairment caused by aging and other aspects of physiological decline with age.
Superoxide dismutase and cancer
Superoxide dismutase, being a key cellular antioxidant, is largely responsible for the elimination of O₂-. Many studies have shown a critical role for oxidative stress in carcinogenesis. Indeed, there is some clear evidence that reactive oxygen species act as an endogenous class of carcinogens, causing mutations in cells. Decreases in the activity of Cu, Zn-SOD and Mn-SOD have been reported in cancer cells. Normalization of superoxide dismutase levels promotes partial reversal of the phenotype of cancer cells. It has been suggested that superoxide dismutase may regulate cancer progression and therefore could be used as a new target for cancer treatment. In addition, it has been shown that Cu, Zn-SOD can be used as a new therapeutic target. for the treatment of multiple myeloma.
Liposomes / superoxide dismutase mimetics have shown promising results experimentally in animal models for cancer prevention. They have also been shown to be safe in the early stages of clinical trials. Supplement-based superoxide dismutase cancer prevention provides another antioxidant-based cancer prevention opportunity. Superoxide dismutase mimetics have been shown to be effective in the treatment of currently incurable castration-resistant prostate cancer in which the expression of superoxide dismutase-2 is severely suppressed. Recently, a potent SOD mimetic, MnTnBuOE-2-PyP (5+), has been shown to enhance carbenoxolone-induced TRAIL-induced apoptosis in most malignant brain tumors.
Superoxide dismutase and diabetes
Increased oxidative stress plays an important role in the etiology of diabetes and its complications. In diabetes, persistent hyperglycemia stimulates the production of reactive oxygen species from various sources. As a result, diabetes usually leads to an increase in the production of active radicals and a weakening of antioxidant defenses. Superoxide dismutase catalyzes the conversion of O₂- to H₂O₂. In conditions of hyperglycemia, endothelial cells produce increased levels of O₂-. Excessive production of O₂- can inhibit glyceraldehyde-3-phosphate dehydrogenase, which is an important enzyme in the glycolytic pathway. This leads to the accumulation of glucose and other intermediate metabolites of this pathway and is shifted to other alternative pathways of glucose metabolism, together with increased production of end products of enhanced glycosylation.
It has been experimentally shown that treatment with superoxide dismutase reduces oxidative stress in the liver in diabetic animals. The superoxide dismutase mimetic has been successfully used to treat diabetes in diabetic rats. Chemically modified superoxide dismutase (carboxymethylcellulose-SOD and polymethylvinylether-maleic anhydride copolymer-SOD) is effective in the treatment of diabetes and has a therapeutic advantage in clinical use. It has been demonstrated that extracellular superoxide dismutase may act as a therapeutic agent to protect against the progression of diabetic nephropathy.
Limitations of the therapeutic use of superoxide dismutase
Due to the instability, high immunogenicity, low cellular uptake and less in vivo circulation of the half-life of superoxide dismutase, their clinical use as therapeutic agents is very limited. For this reason, a large number of superoxide dismutase conjugates have been developed with a longer half-life in the bloodstream, high stability and less immunogenicity. These superoxide dismutase conjugates have shown marked in vivo effects. Administration of free-form superoxide dismutase has several disadvantages, the most important of which is the low accumulation of superoxide dismutase in inflamed areas due to its reduced half-life in the bloodstream and its rapid excretion through the kidneys. To overcome this, superoxide dismutase can be incorporated into either highly loaded conventional liposomes or long-term circulating liposomes (PEG liposomes). A variety of superoxide dismutase mimetics have been synthesized that can be used as pharmaceutical agents for a wide variety of diseases in which native SOD is ineffective. Powerful superoxide dismutase mimetics such as metalloporphyrins, cyclic Mn polyamines, Mn-salen derivatives and nitroxides. have been developed to treat various diseases caused by increased oxidative stress.
GliSODin oral superoxide dismutase
GliSODin is an oral bioavailable form of superoxide dismutase in combination with gliadin. Thanks to the combination with gliadin, the superoxide dismutase in GliSODin is not degraded in the gastrointestinal tract and enters the bloodstream. The source of superoxide dismutase in GliSODin is nutmeg, which is rich in this enzyme.
Read in details - GliSODin.